212-204-0075
info@pyvot.tech
Comparison of Sunshell Biphenyl vs. other manufacturers Biphenyl phase columns using standard samples/basic compounds.
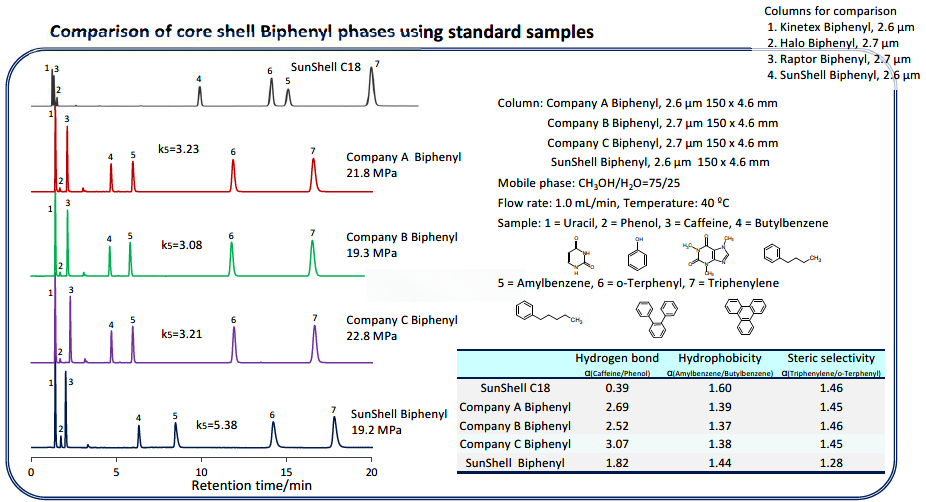
Compared with other company’s core shell type Biphenyl. Biphenyls from companies A, B, and C showed similar separation patterns. It was confirmed that SunShell Biphenyl has higher retention of amylbenzene No. 5 and a larger separation factor (hydrophobicity in the table) for the difference of one carbon between butylbenzene and amylbenzene, and is more hydrophobic than other company Biphenyl. The carbon loading is 5% for SunShell Biphenyl and 7% or more for all other Biphenyls, and the high hydrophobicity of SunShell, which has the lowest carbon content, indicates high-density end-capping. Moreover, the separation of standard samples is very different for biphenyl and C18.
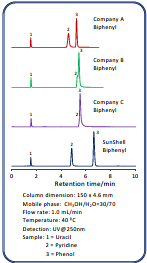
Comparison using pyridine
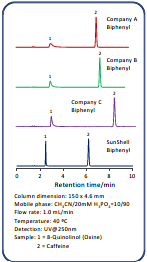
Comparison using oxine
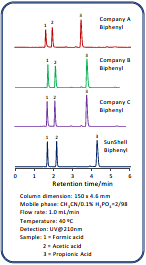
Comparison using formic acid
Pyridine is compound that tends to tail due to residual silanol groups. When methanol is used
as the organic solvent in the mobile phase
rather than acetonitrile, tailing of pyridine is
more likely to occur and the difference
between brands becomes clear.
8-Quinolinol (oxine) is a metal chelating
compound, and if there are metal impurities in the packing material, its peak becomes a
tailing peak.
Although most acidic compounds can be
eluted without problems, formic acid is a typical acidic compound that easily causes tailing. Formic acid is tailed on Company A
Biphenyl.
Comparison of core shell Biphenyl phases using basic compounds
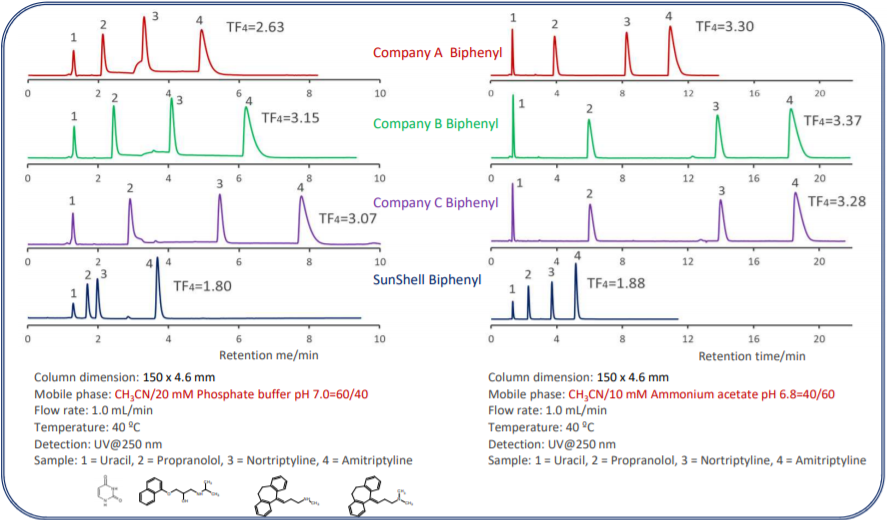
Tailing of basic compounds (amitriptyline) occurs more easily in the acetonitrile/buffer mobile phase than in the methanol/buffer mobile
phase. A mixture of acetonitrile and 20 mM phosphate buffer and a mixture of acetonitrile and 10 mm ammonium acetate was used as
mobile phase to compare with core shell Biphenyls. Biphenyl from other companies had a terrible tailing for basic compounds, and it was
also confirmed that the retention time was increased, which seems to be influenced by the adsorption to residual silanol groups
